Decoding Borax: The Chemical Formula and Its Significance
Have you ever wondered about the magic behind borax, that versatile powder found in laundry rooms and cleaning supplies? Its effectiveness comes down to its unique chemical makeup. This exploration dives into the essence of borax, revealing its chemical formula and the fascinating story behind its multifaceted nature.
The chemical formula for borax is Na₂[B₄O₅(OH)₄]·8H₂O. This seemingly complex string of letters and numbers reveals a hydrated sodium borate compound. In simpler terms, it's a combination of sodium, boron, oxygen, and water molecules arranged in a specific crystalline structure. Understanding this formula unlocks the key to understanding borax's properties and diverse applications.
Borax, also known as sodium borate, sodium tetraborate, or disodium tetraborate, has a rich history dating back centuries. Originally discovered in dry lakebeds in Tibet, its use spread along the Silk Road to the Middle East and eventually Europe. For centuries, borax has been valued for its cleaning properties, used in everything from laundry detergents to pottery glazes.
The importance of understanding the chemical formula of borax lies in its predictive power. Knowing the composition allows scientists and manufacturers to anticipate how borax will react with other substances. This is crucial for developing new applications and ensuring safe and effective use in existing ones. For instance, the borate ions in borax act as a buffer, maintaining a stable pH level, which is essential for its cleaning power and ability to control fungal growth.
One of the main issues surrounding borax is its potential toxicity. While generally considered safe for household use, large amounts of borax can be harmful if ingested or inhaled. Understanding the chemical formula helps to assess the concentration of boron in different borax products and thus determine safe handling practices. It’s crucial to follow product instructions and store borax out of reach of children and pets.
Borax offers several benefits. First, it’s an excellent cleaning agent, its alkaline nature helps to dissolve grease and grime. Second, it acts as a natural insecticide and fungicide. Third, borax is a key ingredient in many slime recipes, providing the characteristic elasticity and texture. These diverse applications highlight the versatility of this compound.
Advantages and Disadvantages of Borax
| Advantages | Disadvantages |
|---|---|
| Effective cleaning agent | Potential toxicity in large doses |
| Natural insecticide and fungicide | Can irritate skin and eyes |
| Versatile applications | Not recommended for internal use |
Five best practices when using borax: 1. Always follow product instructions. 2. Wear gloves when handling concentrated borax solutions. 3. Store borax in a cool, dry place away from children and pets. 4. Avoid inhaling borax dust. 5. Rinse surfaces thoroughly after cleaning with borax.
Five real-world examples of borax use: 1. Laundry booster. 2. Cleaning agent for bathrooms and kitchens. 3. Ingredient in homemade slime. 4. Pest control for ants and roaches. 5. Flux for soldering and welding.
Frequently Asked Questions: 1. What is the chemical formula for borax? Na₂[B₄O₅(OH)₄]·8H₂O. 2. Is borax safe? Generally safe for household use when handled according to instructions. 3. Can borax kill ants? Yes, borax is a common ingredient in ant baits. 4. What is borax used for? Cleaning, pest control, and various industrial applications. 5. Is borax toxic to humans? Can be toxic in large amounts. 6. Where can I buy borax? Grocery stores, hardware stores, and online retailers. 7. How do I clean with borax? Dissolve in water and use as a cleaning solution. 8. Is borax the same as baking soda? No, they are different chemical compounds.
Tips for using borax: Mix borax with water to create a cleaning paste for stubborn stains. Add a small amount of borax to laundry detergent for extra cleaning power. Create a borax and sugar ant bait to control ant infestations.
In conclusion, the chemical formula for borax, Na₂[B₄O₅(OH)₄]·8H₂O, represents more than just a string of characters. It embodies the essence of this versatile compound, from its historical journey across continents to its modern applications in cleaning, pest control, and even children's crafts. While safety precautions are necessary, the benefits of borax are undeniable. Understanding its chemical makeup empowers us to use this powerful substance effectively and responsibly. By appreciating the science behind borax, we can fully harness its potential while ensuring its safe and beneficial use for generations to come. Take the time to research and understand borax’s properties and you'll be well-equipped to utilize its many advantages safely and effectively.
Finding calm with behrs alpine trail paint
Reinvent your outdoor space with behr floor and porch paint
Unveiling the kuronami knife price mystery





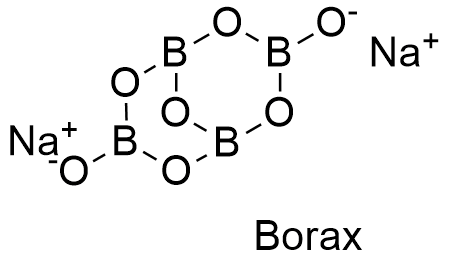




/what-is-borax-where-to-get-608509_FINAL-277034df3e8447dea95cf03285b01155.png)


